Iodine being the least reactive halogen and being a solid is the easiest and safest halogen to handleįluorine at the top of the group is a toxic pale greeny yellow gas, it is perhapsĮlements in the periodic table. The halogens are all very reactive elements and are not found as elements in nature, instead they areįound combined in compounds in rocks and minerals.įluorine, chlorine and bromine are all toxic and corrosive and great care is needed in handling these reactive elements, though Astatine at the bottom of group 7 is a very rare and highly radioactive element its most stable isotope has a half-life of just over 8 hours. The halogens are fluorine, chlorine, bromine and The group 7 non-metals are called the halogens. metals-chemical and physical properties. 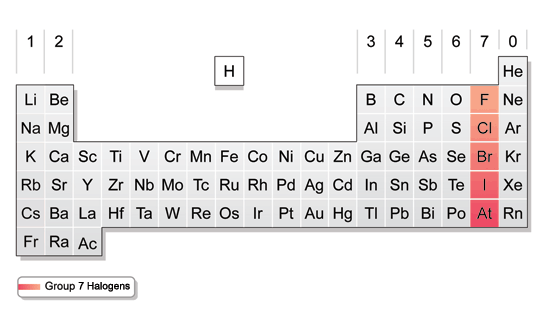
As the stability of halogen acids decreases down the group accordingly their acidic strength increases down group. The acidic strength of hydrogen halides increases down the group.While the order of reactivity of halogen acids is HI > HBr >HCl > HF.The order of stability and polarity is HF > HCl > HBr > HI.The order of reactivity of halogens towards this reaction is F2 > Cl2 > Br2 > I2Īs the size of halogen increases, the H – X bond energy decreases, and thus, the stability of halide decreases and the bond’s polarity decreases. (xv) Halogens directly react with hydrogen under different conditions to produce their hydrides ( Hydrogen halides). They form ionic compounds with group I-A and group II-A elements. However, fluorine does not show a positive oxidation state as it has the highest electronegativity. Group 7A Halogens Common oxidation stateĬommon oxidation state for halogens is -1, but they also show +1, +3, +5, and +7 oxidation states in their compounds. Halogens have high ionization energies, electron affinities and electronegativities. H2SO4 + 8H+ + 8I – → 4I2 + H2S + 2H2O Halogens have irritating odours and they attack the skin.īromine causes burns that heal slowly. It reduces the sulphate ion of sulphuric acid to S-2 ion. (ii) I – ion is larger than Br – ion so it is a stronger reducing agent than Br – ion. (i) Br – ion reduces sulphate ion of sulphuric acid to SO2.Į.g. Larger ions can easily donate an electron and thus can easily reduce other substances.Į.g. The larger the size of the halide ion more is its reducing power. Reducing power of halide ions depends upon their sizes. In this reaction, Cl2 has oxidized I – to I2. (ii) Chlorine water oxidizes KI to iodine and the solution turns brown due to the formation of iodine. Thus they are used as bleaching agents.Į.g. (i) Fluorine and chlorine can oxidize coloured dyes to colourless. Halogens take electrons from other elements and thus oxidize them.Į.g. what is electron affinity Electron affinity Trend Higher is the attraction higher will be the electron affinity. It is the measure of attraction between incoming electrons and the nucleus. Halogens Group VII A Electron affinityĮlectron affinity is the energy released or absorbed when an electron is added to a gaseous atom or ion. Fluorine has the highest electronegativity which is four. Thei r electronegativitiesare high and decrease down the group. Their atomic radii increase down the group. 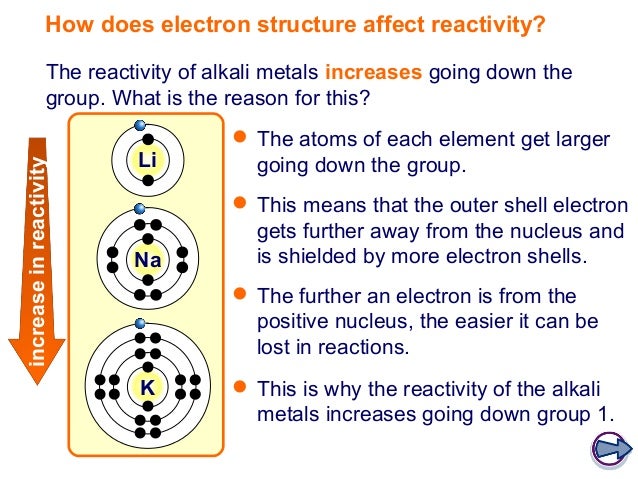
They exist as discrete diatomic molecules F2, Cl2, Br2, I2, At2. Their general valence shell configuration of group 7 (Halogens) is ns2, np5. This group consists of the elements which are given in the following. Halogens belong to group VII-A or group 7 or group 17 of the periodic table. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
